Own developments
Development projects by KONTAKT-Elektro Ltd.
Our company started dealing with fuel cells 15 years ago. This first development result of the company in this field was the production of a 100W fuel cell stack, which was implemented in 2007 in the framework of an INNOCSEKK tender. Next step was to develop the first stack further and changing as many of its components to own developed ones as possible. This was made possible by the GOP tender in 2010 which we won in cooperation with the MTA Chemical Research Institution (later MTA TTK Material and Environmental chemical institute). During the project using the anode catalyst without platinum developed by the MTA KKI as base we were able to produce fuel cell stacks in several versions and sizes. (Katalizátor_MEA). The stacks ranging from 10W to a couple of kW were 10 cm2, 50 cm2, 100 cm2 and 200 cm2 active surfaced open and closed, air and liquid cooling versions were produced (Stack prototypes). The BiPolar plates used for the stacks were graphite based composite plates, part of which were produced by the BME Polymer technology Department (BiPolar plates).
Parallel to the development of the stacks, Kontakt-Elektro Ltd also started developing fuel cell based facilities. Our first facility was a 300W portable power supply tool, that we developed specifically for the power supply of small electric boats. Besides the case we also installed a metal-hybrid hydrogen storage built into a portable frame which is able to supply the device with hydrogen for about 6 hours (portable power supply).
We launched a bigger project in 2010 in the topic of equipment development, for which we won the support of Gábor Baross tender together with the Faculty of Engineering and Information Technology of the University of Pécs. The aim of the project was to build the prototypes of stationary and portable applications until 5kW performance based on fuel cell. To realize the project, we were planning to use the market stack, this is how we got to know the Canadian Ballard company, who is and has been a leading producer of fuel cells since its launch (power sources based on Ballard stack).
The first prototypes which was based on the Ballard stack was a bicycle powered by a 400W fuel cell (fuel cell bicycle). After this, we built a 3kW uninterrupted power supply designed for 48VDC systems, which a similar performance, accumulator based hybrid power source facility followed. Later this hybrid power supply was the base of the power supply of the fuel cell boat which was introduced in 2013 on the Boat Show of Balatonlelle (FC ship). Besides the Ballard FCgen-1020ACS air cooling stack family, we used the company’s FCgen-1310 types of liquid cooler stack as well in two other facilities: we made a 4.5kW uninterrupted power supply, and a similar performance, cogeneration (capable of heat and electricity production) facility. Besides the items listed above, we advised the Orca racing car designed for the international ECOMarathon competition built by the university, the first version of which was supplied by the 1.2kW stack built by our company.
Besides the fuel cell facilities, we also deal with other connecting hydrogen technologies as well. In 2012, we built an electrolyte facility supplied hydrogen recharge container which was powered by solar panels, with which we were able to fill hydrogen to cylinders until 300 bar using explosion secure, pneumatic power pressure enhancing from the 15bar buffer containers (Hydrogen recharge station powered by solar panel). Developing the previously mentioned concept further in 2013, we built an autonomous energy container, which operates consumers using electricity and solar panels, using energy surplus it creates hydrogen which they store in a 30bar buffer container, where in case of power outage electricity is produced by fuel cell above the need of the power solar panels can supply (Energy container). To demonstrate similar power supply scheme, we built a facility that rolls on wheels, in which we integrated electrolyzing facility and fuel cell, with inverter and solar panel connecting possibility (hydrogen energy container facility).
Over the years we have gathered experience, which moved our focus form developing fuel cell stacks to purchasing facilities and developing them further, which was less time-consuming, gave us the opportunity to put it on the market faster, though the knowledge we gathered while developing the stacks and their components became invaluable when building the purchased stacks further, by providing the perfect operating conditions and designing the BOP (Balance of Plant) system necessary for operation.
After the tender projects, we saw business opportunity in mainly the installed applications, more specifically the uninterrupted power supplies, so we continued developing in this field. In 2014, we finished building a modular, rack based uninterrupted power supply which we were able to sell on the market (FC-UPS 2500 – introduction).
After the upturn of fuel cell technologies in 2017 we rebuilt our contact with a cell producer company, and were able to purchase a 15kW, modern fuel cell module from Hydrogenics, using which we could aim for the performance range above 10kW. Our first equipment was a 15kW power supply which rolled on wheels, which was perfectly capable of providing energy for concerts, festivals, and movie productions due to its noise strain and zero emission (FCPS-15 – introduction).
Other area where we used the module of Hydrogenics was a little ship designed for recreational purposes which uses a 15kW electric engine and an accumulator-fuel cell hybrid power supply system, where the hydrogen supply is rechargeable using a 300bar composite cylinder storage system (H2-evolution yacht). This yacht was introduced in September 2020, on the Boat Show of Balatonkenese.
Our plan for the future is to develop a bigger ship based on the 15kW small ship (Hydrogen based electric yacht), and the prototype of a waste collecting vehicle (Municipal and commercial vehicles).
You can download the information sheet about our own developments by clicking on the following links:
- Catalyst MEA
- Stack prototypes
- BiPolar plates
- Portable power supplies
- Power supplies based on Ballard stack,
- Fuel cell bicycle,
- Fuel cell ship
- Solar panel powered hydrogen power station,
- Energy container
- Hydrogen energy storage facility
- FC-UPS 2500 – introduction
- FCPS-15 – introduction
- H2-evolution yacht
- Hydrogen powered electric yacht,
- Municipal and commercial vehicles
Fuel cell applications
Many options of fuel cell applications are known today, more and more opportunities are expected to be discovered in the future, as the technology becomes widely available, the prices decrease due the gradually newer and more compatible technical parameters.
The fast improvement was inspired by the strong need for utilization in transportation, so mostly the PEM type fuel cell used in vehicles was the most involved, but this interest has extended to other types as well and speeded up the development of cells of other fields.
Fuel cells can be categorized based on the type of the electrolyte, since they mostly determine the cells’ attributes. Therefore, utilization can also be connected to one or two types mostly. The 6 main cell types can be utilized in the following areas:
Installed applications
Installed applications just like their names suggest mean fix, installed facilities. However, every application is listed in this category non-portable and non-transportation related items, the aim of which is not an exact consumer (e.g.: electric engine) that makes part of the facilities, but general production of electricity for any external user. In many applications, besides the production of electricity they make use of the temperature loss of the cell as well, thus making combined power (co-generation) which they also call joint energy production as well.
Installed fuel cell systems can be divided into the frequency and continuity of their operation, which is usually in connection with the life span of the cell:
- continuously operating, mostly energy supply applications – this includes the purely energy production or connected energy production (CHP) or recently triple energy production (CHHP, Combined Heat, Hídrogen & Power) purpose technologies with a lifespan of 20-30,000 hours stack
- temporary operation (e.g.: uninterrupted (UPS), or emergency power supply stationary facilities with the lifespan of 3-5,000 hours stack
Regarding performance, there are a lot of types of installed fuel cells available. Based on the non-official categorization the following major categories exist, of course there are inter performances, and modular structures that can be installed based on specific needs (https://www.hfc-hungary.org/energiatermelo-telepitett-alkalmazasok/):
- Domestic fuel cell systems: ~0.3 – 5 kWu,
- Commercial/industrial scale fuel cell systems: ~100 – 500 kWu,
- Utility scale fuel cell systems: from ~1 MWe to more than ten MWe performance. The biggest currently functioning fuel cell system has the capacity of 59 MWe. It is located in South Korea and it consists of 2.8 MW unit performance based MCFC fuel cell stack.
Besides installed facilities, similar purpose (listed under stationary applications), but portable solutions (container, trailer etc.) as well. These usually cover smaller performance (5-100 kW) needs. Toyota’s idea is an interesting concept in this matter, when it comes to energy production use of their vehicles: they sold Mirai in some countries with installed sockets, through which you may sell electricity which is produced by the vehicle’s fuel cell. It is able to supple a household with electricity in case of need. Another version of this concept is the fuel cell buses that form a power station which may be used in events of catastrophe or places with weak infrastructure to provide temporary electricity for the area.
Fuel cells spare systems
At the systems where availability – for example data centers, telecommunication stations – is highly important power outage may cause financial loss. Traditional, accumulator packages and/or diesel generators as power sources do not prove to be stable energy sources.
Hydrogen based power supply systems offer better solution than tradition power sources:
- Easily scalable performance, flexible operational time
- Reliable operation, low maintenance and remote control
- Long lifespan, low service need
- Silent, environmentally friendly operation
- Wide operation temperature range
(Picture source: https://www.hydrogenics.com/hydrogen-products-solutions/fuel-cell-power-systems/stationary-stand-by-power/telecom-data-centre-backup-power)

Hydrogen mobility
Hydrogen used as fuel can remove carbon dioxide from transportation if we use sustainable energy sources. In this case, the biggest benefit is zero CO2 emission and low environmental impact since its only byproduct is water.
Aerial transport

When it comes to aerial transportation, hydrogen is regarded as power source, since it has been used at space travel for a while. Fuel cell modules supply the planes’ electric systems by providing emergency generator or supplementary power supply. They make it possible for planes to start their gear and move on the runway.
There are small sized drones that fly using electricity powered by fuel cells. (https://www.hes.sg/hycopter) An American company cooperating with the Hungarian Genevation Aircraft to build a passenger drone that is powered by fuel cell. (https://www.youtube.com/watch?v=cPfBhZYKm2E)
In the future, zero emission will be the aim at aerial travel as well: for now, some bigger companies are testing hydrogen powered, altered gas turbines. (https://totalcar.hu/magazin/2020/01/29/uzemanyagcellas_szemelyszallito_dron_keszul_magyarorszagon/)
Water transportation
Hydrogen is also used as deck power source at water transportation, it is still in its early stages as drive in small passenger ships and ferries.
This is the area where Kontakt-Elektro Ltd is also actively participating: it introduced its first fuel cell powered ship (2.7kW) in Balatonlelle in 2013 on Balaton Show. Later in 2020 in Balatonkenese, a 15kW yacht that is supplied with hydrogen storage and can be filled from the coast. (H2-evolution jacht.pdf).

Railway transportation

Since the world’s first fuel cell train, the Coradia iLint which was produced by Alstrom in 2018, the interest towards the technology being applied in railway has grown gradually. Since September 2020, Coradia iLint has been present in Austria, and they have also signed a contract with the Dutch railway company.
Besides Alstom, Siemens has also started developing its own fuel cell train, which is based on the company’s electric train, Mireo Plus.
Heavy duty vehicles, buses

Air pollution is a serious health concern, and the transport sector takes a huge part in it. Zero emission in these vehicles has never been more important.
Comparing battery-electric trucks, fuel cell technology offers longer range, performance, load capacity, and fast recharging.
A manufactured fuel cell truck has the following attributes:
- Charging time: 8-20 minutes
- Range: ~400 km.
- 190 kW performance.

In urban public transport, many expect the conquest of fuel cell power. Many bigger cities saw demonstrations and projects for operating fuel cell powered buses on an experimental basis.
You can find more information about European projects on the following website: https://www.fuelcellbuses.eu/. Since the launch of the first project in 2010, the participating buses have collected over 11 million kilometers worth of performance.
Passenger cars
Hydrogen fuel cell can be an alternative solution to the only battery-powered, zero emission passenger cars.
Though hydrogen is a clear source of energy, with beneficial chemical attributes, it hasn’t yet become a popular source of energy in transportation. Based on the experiences of the past few years, the technology will be more and more in the center of passenger cars as well.
Nowadays, there are plenty of practical experiences available about fuel cell based cars. This is due to the fact that some bigger manufacturers started producing it since the middle of 2010, which provide the same quality as their traditional, internal combustion counterparts. The price of a middle-sized fuel cell car is still much lower, than the traditional internal combustion competitors, but with mass production this number will decrease even further.
Toyota Mirai or Hyundai Nexo (earlier ix35) models already possess exceptional fleet in Western European countries. Toyota is planning to launch its Mirai model in Hungary in 2021, which might encourage building more hydrogen refill stations which have been in store for quite some time.
About fuel-cells
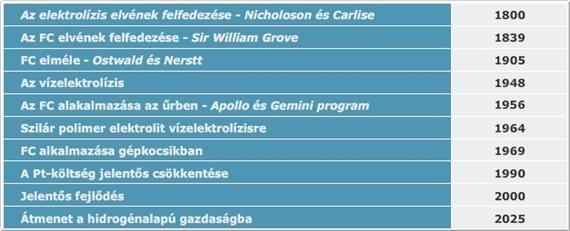
History of fuel cells
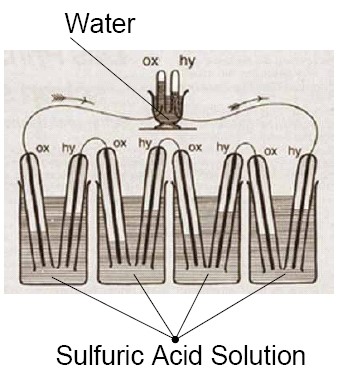
Sir William Robert Grove (1811–1896) noticed in 1838 that if he electrolyzed water, a current in the opposite direction would flow after the applied current was turned off. This current was created by the hydrogen evolved at one platinum electrode while the oxygen generated at the other is reduced by reversing water decomposition. Grove took advantage of this discovery, and designed his first fuel cells, which – to distinguish from other elements among which the reaction between metals and their compounds provided electricity – named is as a gas element. The gas element consists of two platinum electrodes that are immersed in sulfuric acid solution. One electrode extended into hydrogen and the other into oxygen tank. Grove also realized that the levels of the solutions was rising when current is flowing between the two electrodes. This indicated the depletion of hydrogen and oxygen. Even though fuel cell was discovered by Grove, its function was completely explained by Ostwald. He introduced the terms electrode, ion and the electrocatalyst. He also introduced semi-cell reactions, namely that hydrogen oxidizes, oxygen is reduced on catalytic surface of Pt. The electrons produced during the redox process can be made to use, while hydrogen ion leads charge in the electrolyte (in case of a Grove cell). He also introduced the terms gas electrode and triple-phase boundary which still make an important element of the research of fuel cell optimalization.
The gas element that Grove discovered worked with clear hydrogen, and since the electrode was plane electrode, he had to use great surfaces. However, Mond and Langer produced hydrogen using carbon in 1889 (synthesis gas). In their fuel cell they used electrodes with three dimensional pores, using which they were able to produce 65 A/m2 on 0.73 V. So basically, they had everything they needed by the beginning of the XXth century which we use today in fuel cells. Ostwald was right in believing that thermal machines will be replaced by electrochemical processes due to their higher effectiveness and that hydrogen will become a universal fuel, and fuel cells will become important energy source units in the XXth century. Fuel cells were first used not in energetics, but in space research. However, this is not how the story began…
Francis Bacon read a German article in 1932 which was about a process used to dissociate water with unnecessary electricity the night mode and the hydrogen produced this way would be burnt in internal combustion engines as fuel. Bacon thought they would be much more effective if they produced electricity the electrochemical way. His first suggestion “Energy storage battery” was about storing electricity, but he didn’t receive any sponsorship. That was when he decided he was going to make his first cell. He didn’t use platinum, probably due to its high price, and used activated nickel net in wet NaOH tincture. He separated the electrodes using salamander wool. He wanted to increase the activity of nickel above 100°C temperature and high pressure (210 bar). since steam machines were used at the time, isolation wasn’t an issue in this pressure and temperature territory. First, he designed a reversable cell, but he finally built a two-cell system, he used one of them for water dissociation and the other for producing electricity. Finally, the cells worked on 200°C and 42 bar pressure. The highest performance of the fuel cell was not higher than 100mA/cm2 (today a better cell’s performance is 1500 mA/cm2) and it had plenty of issues. Finally, he received a pored nickel piece from a secret research, using which he was able to reach the desired density. His next problem to solve was the guaranteeing of stable surface between the hydrogen, oxygen, and electrolyte. For this purpose, multi-pole electrodes were created, which contain bigger poles on the gas side and smaller ones on the electrolyte side, so they can punctually control the entering depth of the gases.
They created the instable diaphragm by applying slightly lower pressure difference (0.14 bar) between the gases and the electrolyte). The next task was to increase the lifespan of the cell. The oxygen electrode quicky oxidized, it was dented then collapsed. They realized if they used oxide layer on the surface then it would protect nickel from further corrosion. Then they experienced other problem, the layer of oxide acted as electrical isolation. They could eliminate this problem if they topped the layer with lithium. It seemed that with the elimination of the oxygen electrode problem, the cell would work fine, but then the hydrogen solution lost its activity. They realized it was caused by the catalyst poison leaving the fittings. To prevent this, they added Teflon to the fittings. So, by the middle of the 50s, they created the first operating system which supplied 150W: 230mA/cm2 density, 0.8V cell pressure. Shortly after this, this discovery reached the USA, and Prett&Whitney spent 100 million dollars and employed 1000 engineers to make a useful product from the demonstrational model.
The Bacon-cell is used perfectly in space research, since it provided electricity, heat, and water at the same time for the astronauts, where they had a constant supply of hydrogen and oxygen. Later there were several attempts to make use of the Bacon cell in industries, but it didn’t bring about much success.
Introduction and categorization of fuel cell types
They created the instable diaphragm by applying slightly lower pressure difference (0.14 bar) between the gases and the electrolyte). The next task was to increase the lifespan of the cell. The oxygen electrode quicky oxidized, it was dented then collapsed. They realized if they used oxide layer on the surface then it would protect nickel from further corrosion. Then they experienced other problem, the layer of oxide acted as electrical isolation. They could eliminate this problem if they topped the layer with lithium. It seemed that with the elimination of the oxygen electrode problem, the cell would work fine, but then the hydrogen solution lost its activity. They realized it was caused by the catalyst poison leaving the fittings. To prevent this, they added Teflon to the fittings. So, by the middle of the 50s, they created the first operating system which supplied 150W: 230mA/cm2 density, 0.8V cell pressure. Shortly after this, this discovery reached the USA, and Prett&Whitney spent 100 million dollars and employed 1000 engineers to make a useful product from the demonstrational model.
The Bacon-cell is used perfectly in space research, since it provided electricity, heat, and water at the same time for the astronauts, where they had a constant supply of hydrogen and oxygen. Later there were several attempts to make use of the Bacon cell in industries, but it didn’t bring about much success.
Introduction and categorization of fuel cell types
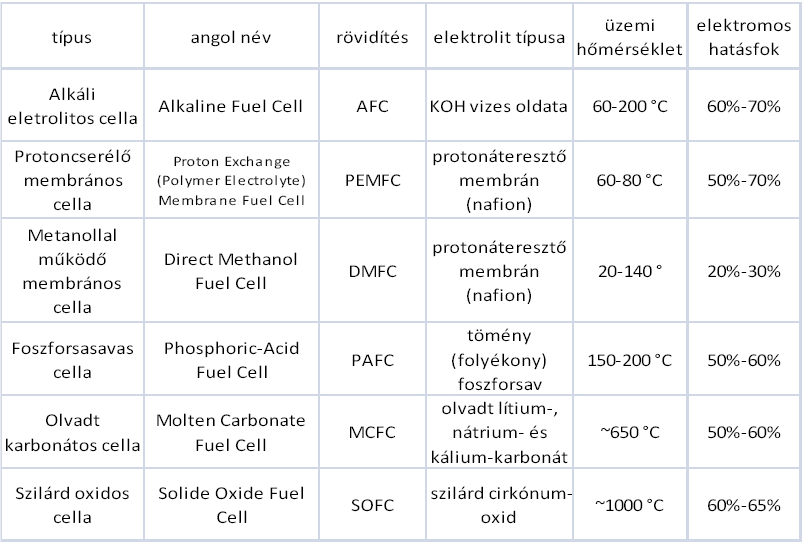
There are several ways to categorize fuel cells based on different criteria. Based on its function temperature there are two main types that we distinguish:
- Low-temperature fuel cells
- High-temperature fuel cells
The most widespread categorization is based on the applied electrolyte, this is what we will use here on out.
The alkaline fuel cells (AFC) represent the most elaborated technologies from the fuel cells. They have been used since 1950, mostly NASA Apollos and Space Shuttle programs. These types of cells provided electricity for the functioning of some tools and drinking on the space shuttle. These types of fuel cells are named after its inventor, Bacon-cells.
In case of the alkaline electrolyte cells, the charge emitting particle is hydroxide (OH-) which moves from the cathode to the anode, where it reacts with the hydrogen which creates water and electrons.
Operating conditions:
The type of electrolyte: e.g.: 30% water potassium hydroxide solution
Operating temperature: below 80°C
Electrical efficiency: 60%-70%
Reactions:
Anode: 2H2 + 4OH- → 4H2O + 4e-
Cathode: O2 + 2H2O + 4e- → 4OH-
Overall: 2H2 + O2 → 2H2O
Advantages:
- One of the most electrically efficient cells.
- Its production is very cheap, because it functions with many different electrolytes.
- It has a considerably low operating temperature.
- Fast diffuse.
Disadvantages:
It is very sensitive to carbon dioxide, carbon monoxide and methane as they can react with the electrolyte and decrease the efficiency of the fuel cell. Its best place is in isolation from the outside world where the above mentioned gases are not threatening. For functioning, it needs clear hydrogen and oxygen.
Application areas:
- Submarines
- Ships
- Armaments industry
Molten Carbonate Fuel Cell (MCFC) belongs to the high operating temperature cell category. Due to the high operating temperature, the fuel cell can be operated directly by natural gas. It was developed in the mid-1960s, and since then they had the greatest achievements in regard to performance and lifespan growth. These cells work differently from other cells. As electrolyte they contain melted carbonate salt, usually as a combination of two carbonates. The two most usual combinations are lithium carbonate and potassium carbonate.
High operating temperature is necessary for the melting of the electrolyte and to ensure adequate ion transmissivity. After melting, the electrolyte is able to conduct carbonate ions (CO32-). These ions move from cathode to anode where merging with water they produce water, carbon dioxide and electrons. The electron passes through a different external current produces electricity and heat while arriving back to the cathode.
Operating conditions:
type of electrolyte: melted lithium, natrium and potassium carbonate
Operating temperature: above 600°C
Electric efficiency: 50%–60%
Reactions:
Anode: CO32- + H2 → H2O + CO2 + 2e-
Cathode: CO2 + ½O2 + 2e- → CO32-
Overall reaction: H2 + ½O2 + CO2 (cathode) → H2O + CO2 (anode)
Advantages:
- Due to high operating temperature, fuel reformer is not needed. These fuel cells are often called internal reformer cells.
- The high operating temperature allows efficient heat usage.
- Cheap ingredients.
Disadvantages:
- Sensitive to corrosion
- Slow diffuse
- Difficult to control the flow of carbon dioxide.
Application areas:
- Cogeneration power stations
- assistant power sources
Phosphoric acid fuel cells (PAFC) were the first to appear on commercial markets from all fuel cells. This type was developed in the mid-1960s, and after the first decade it was in line for the first sales. In comparison to other fuel cells this proved to behave in a more stable way, it reached higher performance and besides all this its cost was much lower. Electrolyte in these cells consists of phosphoric acid (H3PO4) fully. Since the phosphoric ion lead on low temperature is weak, so the operating temperature is quite high, often exceeds 200°C. Its operation is mostly similar to the proton-exchange membrane fuel cells. Therefore, the reactions of anode and cathode are fairly similar as well.
Operating conditions:
- type of electrolyte: dense, liquid phosphoric acid
- operating temperature: 150-220 °C
- Electrical efficiency: 50%-60%
Reactions:
Anode: 2H2 → 4H+ + 4e-
Cathode: O2 + 4H+ + 4e- → 2H2O
Overall reaction: 2H2 + O2 → 2H2O
Advantages:
- The high operating temperature allows efficient heat recovery.
- Unsensitive to carbon dioxide and carbon monoxide.
- Stability
- Simple structure
Disadvantages:
- Big size
- Platinum catalyst is needed.
- Difficult to diffuse (phosphoric acid is solid below the temperature of 40°C)
Application areas:
- Energy supplies of buildings
- Power stations
- Armaments industry
Proton Exchange Membrane Fuel Cell (PEMFC) might be the most suitable to replace the currently used diesel and petrol engines in the future of transportation. First, it was used by NASA in the Gemini program in the 1960s. The material of the electrolyte in these cells is solid polymer membrane (thin plastic layer of film). What makes this polymer unique is that it transmits protons once its wet, but not the electrons.
The hydrogen flooding in from the anode falls apart on the surface of the catalyst into protons and electrons. The protons move toward the cathode through a membrane, the electrons reach it through an external current, while producing electricity. The electron arriving at the cathode merges with oxygen and the hydrogen arriving from the membrane and they produce water.
Comparing it to many different fuel cells, it possesses much better energy and performance parameters. One of its specialties is that its operational temperature mainly depends on the material of the membrane. One of the most frequently used material as a membrane is Nafion® which requires a lower operating temperature, while the polybenzimidazole membrane might exceed 200°C as well.
Operating conditions:
Type of electrolyte: proton transmitting membrane.
Operating temperature:70–220°C
Electrical efficiency: 50%-70%
Reactions:
Anode: 2H2 → 4H+ + 4e-
Cathode: O2 + 4H+ + 4e- → 2H2O
Overall reaction: 2H2 + O2 → 2H2O
Advantages:
- Efficient
- Due to solid electrolyte, it is not sensitive to gravitation.
- Fast diffuse
- Long lifespan
Disadvantages:
- Difficult to control (the electrolyte needs to be wet)
- Due to the lower operating temperature the heat consumption use is also low.
- Due to its precious metal content, it is expensive.
Application areas:
- Automobile industry
- Armaments industry
- Portable power sources
- Power stations
Structure of cells
- two electrodes (anode and cathode)
- electrolyte or membrane
- catalyst (platinum or its compounds)
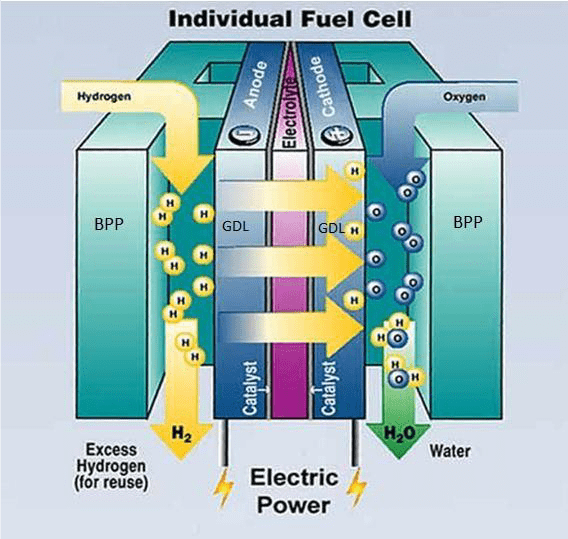
The structure of fuel cell is illustrated on image 3. Anode is the name of the electrode that oxidizes, or the fuel flowing in here passes on electrons, while cathode is place for reduction (taking on electrons). The electrolyte isolates the two electrodes, preventing the fuel and the oxidization material to mix. Nowadays, the proton transmitting membrane cells are more popular, where the task of the membrane is to let hydrogen ions pass through the membrane. The electrons get from the anode to the cathode through a metallic lead connecting the electrodes. The gas entry and the homogenic distribution is aided by graphite gas leading plates (BPP- BiPolar Plates) on the side of the anode and cathode and graphite or textile plates (GDL – Gas Diffusion Layer). By implementing a consumer (e.g.: electro engine) the electrons can be put to work, the measure of which depends on the potential difference between the two electrodes and the measure of transmitted charge. The structure and operation of the cell can be observed on the image below.
The electrolyte also acts at an electric isolation, but it is a perfect transmitter of ions as well. The two important differences are the operating temperature and the material of the electrolyte responsible for the transmission in the types of fuel cells.
The operation of PEM cells
The hydrogen passing through the anode is selected by the platinum catalyst into hydrogen atoms. After this the H+ ions move to the cathode through a membrane. The membrane is often called proton exchanger as it only allows the hydrogen ion pass, so basically the protons, while not the negative charged electrons. The electrons can move to the cathode through an external consumer. The oxygen molecules led to the cathode are degraded with the help of a catalyst, which form water emerging with the electrons flowing from anode and the hydrogen moving in through the membrane.
The reaction in the electrodes:
Anode: 2H2 → 4H++4e– (3.1)
Cathode: O2+4H++4e– → 2H2O (3.2)
A membrane is usually a solid plastic plate, which is usually covered in platinum. Platinum is the best catalyst, but it is expensive, so the platinum or its compounds have to implemented into the surface of the membrane as little as possible.
It is important that moisture in the system should be at adequate level because the ionic conductivity of Nafion type membranes deteriorate in case of low or too high moisture. The newest inventions are the dry operational membranes. The control of moisture has an effect on osmosis pressure because this determines the flowing direction of moisture, which has to be the same as the flow of the protons, otherwise it prevents it. On the other hand, the byproduct of the reaction is water so its diversion is also important. It is advised to keep the temperature between 80 and 90 °C, but at this temperature the reactions take longer time. The control of temperature includes heating and cooling, since if the temperature is too low, the water can freeze into the membrane which also prevents the flow of the protons.
Life span of fuel cells is highly influenced by the cleanness of the fuel. The platinum catalyst can easily be polluted by the CO flowing in the fuel, so it has to meet certain criteria. The allowed carbon monoxide content is below 100 ppm.
Source: Mayer Zoltán és Kriston Ákos – Hidrogén és metanol gazdaság (https://regi.tankonyvtar.hu/hu/tartalom/tamop412A/2010-0017_31_hidrogen_es_metanol/ch01s04.html)
Hydrogen Energetics
80% of the global primary energy need is made up of fossil energy sources, besides which nuclear energy as well as sustainable energy sources are fairly limited. Consumption is expected to grow in the future, therefore it is forecast that the high ratio of energy sources will not be sustainably secure in the long term, or even in medium term.
Risks and limitations related to fossil fuels are manifested from several sides and their effects are significant even if we they are individually regarded. Maybe the most known of these is the source limitations, which suggest the obvious end of supply, and which we introduce in more detail under the chapter on oil peak.
In recent years, sink-side limitations have risen to similar aspect to the source-side limitations. By using fossil fuels (most frequently: burning) poisonous gases are emitted, mostly the green house gas carbon dioxide which can not be absorbed by the Earth’s atmosphere, more specifically its ecosystem (which in this case can be regarded as absorber), at the same rate as it is emitted. One of its significant signs is the estimated 30% growth in the atmosphere’s carbon dioxide concentration in the last two to three hundred years due to human activity, the result of which – still not known in every detail, but – causes the great challenge of global climate change.
Regarding the use of fossil fuels, a negative tendency is seen throughout the European Union and specifically in Hungary, that we are exposed to foreign import when it comes to energy sources. In Hungary, the ratio of production and import changed from 42-58% in 2000 to 38-62% in 2010, which means our energy dependence has grown in the last decade and continues to remain higher than the EU average.
Nine thirds of the imported energy sources is made up of natural gas, crude oil, and its different derivatives. Due to aging population, just like Hungary’s, this outflow of income (so the income paid for imported energy sources) poses a great threat. In the case of Hungary this is quite significant: considering the world market prices it means 14 percent of Hungary’s total world export revenue, which is spent on energy import, furthermore – as we saw above – the tendency of energy dependence has been on the rise.
The role of electricity as a secondary energy source has become more important in the developed countries, de but tendency can already be observed in the developing countries. The reason for this is that electricity is considered the most valuable energy type, since it can be used for many purposes comfortably, at its application point it does not pollute the environment (however on the production side it causes great pollution), at the application point it can effectively be transformed to any given energy form that we need, and it is transportable fairly easily. The statistics show that the share of electricity used in total energy consumption shows an increasing tendency, in Hungary as well as internationally.
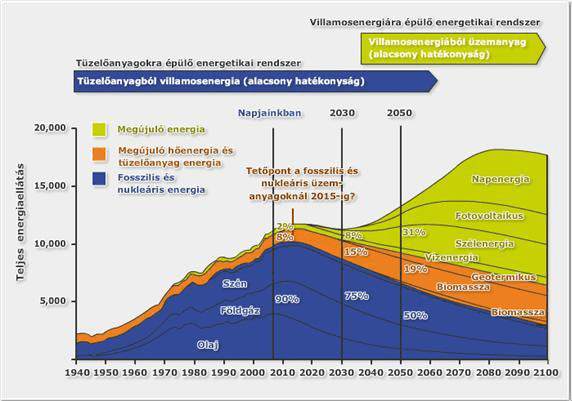
According to 1. image the general future tendency, by the end of the first decade of the 21st century – the current – “fuel to electricity concept” (which involves the alteration of fossil fuels to electricity), will see a temporary phase, and then will gradually shift to an “electricity to fuel” system. In case of the latter one, the direct electricity production will occur at a much higher ratio, and for example in the field of transportation electricity will be used either directly within transportation sector (accumulator based solution) or – due to the lack and problems of battery technology – intermediate energy sources e.g.: through hydrogen, which the “electricity to fuel” expression implies.
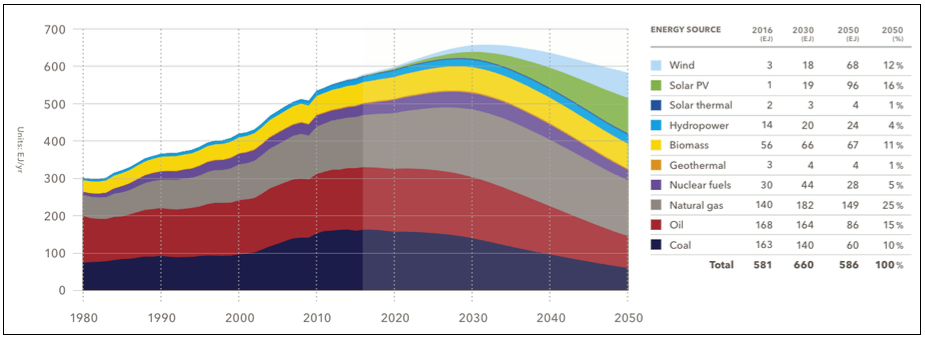
(Note: The previous graphs of 2008 and 2010 and the peak of fossil and nuclear fuel forecasted by 2020 did not realize, while the forecasts of today put the date to between 2020 and 2030 – see 2. image)
In the future, an energy system like this, where electricity dominates, hydrogen, as an intermediate energy storage medium and energy carrier, may possess important value, due to the fact that the so-called fuel cell technologies are able to convert hydrogen to electricity with high efficiency. With a little exaggeration, but hydrogen and electricity could become synonym expressions in an illustrative comprehension. Electricity is a special product mostly because it cannot be stored – at industrial scale – therefore in an electricity dominated system (to maintain an electricity production and consumer balance) control is becoming a more important question, so hydrogen may pay an important role in the future of EPS (Electric power system) when it comes to the control tasks.
Hydrogen is the simplest chemical element, the first element of the periodic table (symbol: H). It was discovered by Henry Cavendish 250 years ago. Its name “Water producer” was created by Antoine Lavoisier from the combination of the Greek hydro (ΰδωρ = ’water’) + genes (γεννώ = ’genes’) words. In natural state it is a colorless, odorless, tasteless, and quite flammable diatomic gas, with high thermal conductivity. Its density is approximately14 times smaller than that of natural air and possesses numerous diffusivities. Hydrogen is the most abundant element of the materials of the universe around 75% (m/m) is hydrogen, but it is also quite popular on Earth: considering the percentage of atoms it ranks 2nd among all the other elements, regarding its mass percentage it ranks 9th. On Earth, hydrogen does not appear in elemental of diatomic state of gas, but mostly in compounds; it is present in water, and in almost every organic compound or biomass. Molecular hydrogen can only be found in smaller amounts in the top layers of the atmosphere.
Its attributes are favorable from many aspects: non-toxic, non-corrosive, non-carcinogenic, non-greenhouse, non-radioactive, its accidental appearance in the environment does not cause any permanent environmental damage.
So, hydrogen is a very common element, and theoretically it is available in “unlimited” amounts, however, only as a compound, which can be retrieved using considerable amount of energy (e.g.: decomposition of water 28kJ/mol). Its production is possible using a lot of methods, in a decentralized way meaning in smaller steps, it can be produced onsite, and – in fuel cells – it can be used highly efficiently. This widespread and big quantity of geographical occurrence is completely opposite to the currently used fossil fuels, which – mostly for example oil – focuses on certain regions, which are not rarely politically instable regions. Hydrogen dissolves in water only fairly, but quite well in metals (palladium, platinum, nickel).
Hydrogen cannot be regarded as a new discovery, not just because of its 250 year-old original discovery, but also due its almost 100 year-old industrial use as well.
Hydrogen has been used, for mostly industrial purposes, but currently and in a couple of years’ time, we will see its everyday, wider and extended use for energetic purposes. It is obvious based on the previous experiences, but maybe it is still important to emphasize, that hydrogen is not a power source, but it is a secondary energy source (similarly to electricity, the secondary “nature” means that it can be produced by mainly using primary energy sources e.g.: natural gas).
For the sake of entirety, it is worth mentioning, that the use of hydrogen for energy purposes was known even in Hungary decades ago, as hydrogen was a high ratio component (~35% V/V) of the so-called “urban gas”. The residents used urban gas for heating and cooking until the1960s. (Another important yet dangerous component of urban gas is carbon monoxide.)
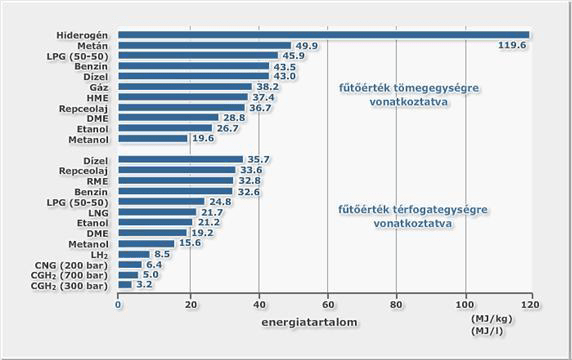
The heating value of hydrogen is the highest from among the known elements (120MJ/kg) regarding its mass unit index, but since the density of hydrogen is quite small, energy density to capacity unit is quite small. Energy content (heat value) of hydrogen regarding its mass and density unit is illustrated on image 3 below in comparison to other applied energy supplies.
Calorific value of Hydrogen considering its mass unit is three times of petrol’s, about 2.4 times of natural gas and six times that of methanol; but its calorific value regarding its unit volume it is third of petrol, about 42% of natural gas and about half of methanol.
Production of Hydrogen using sustainable energy sources
Wind power plant
Traditional (current) hydrogen production methods:
- On the one hand, directly or indirectly, they are all quite polluting for the environment. Directly in case during production there is emission of pollutants. Water decomposition with energy retrieved from electric network is polluting, if in the mix of energy the fossil fuels are dominant, because water decomposition is not polluting, and later the use of hydrogen either, but later producing electricity depending on its method could be polluting (this is what indirect polluting of hydrogen means).
- On the other hand, they rely on finite fossil energy sources, so it would have the same result, if we base bigger amount of hydrogen production on for example natural gas. (Even though in the early stages of hydrogen energy it will be an acceptable method, or it could be; because in the beginning the existence, operation and demonstration of hydrogen technologies would be a big step forward.)
Production methods can be categorized from several aspects; in this case we use the energy source. In this chapter, we introduce hydrogen production using wind power primarily and, in more detail, this is the most mature production method that is based on sustainable energy for which every necessary tool can be purchased on traditional platforms and there are a few of these in operation in the world.
Kinetic energy is converted into mechanical (rotation) energy with the help of wind, then into electrical energy using a generator. With the produced electricity or at least with a part of it, hydrogen can be produced with water decomposition. This production method is called wind hydrogen, which translates as “szél-hidrogén” intro Hungarian; these systems have low (almost zero, to be more exact only at the time of the production of the tool) emission of pollutants.
One disadvantage of wind turbines – more specifically weather dependent sustainable energy sources – that electricity production depends on meteorological parameters (in this case of the speed of the wind) resulting in different amounts of production, while the EPS balance has to be maintained all the time. It is typical for the production capacity variations that the maximum upward and downward change of the external performance after 10 minutes in case of individual power plant farms is the 85-95% of the installed performance. In case of wind farms located across the country and in higher amounts the performance shows much lower fluctuation. This is the changing attribute of the electricity produced by wind turbines that can be evened out in case of hydrogen production, when the EPS is not able to take in the energy produced by wind plants. Based on this the following alternative solutions exist for hydrogen production using wind power:
“Island operation” when the water decomposition system is installed right beside the wind turbine (or appropriate hydrogen storage capacity), as well as the water dissolution performance to be able to take on energy from the wind plant. This type of island production system is technologically possible, from an economic perspective – disregarding the special exception – won’t be viable, mostly due to the high cost.
In case of “Combined production” the wind power plant primarily produces electricity, and the electricity would only go to the water dissolution system and produce hydrogen in case the EPS is unable to take in the electricity produced (e.g.: during the night period). In systems like these two products are produced: electricity and hydrogen. In some – rare – cases, when the water dissolution system produces oxygen on the other electrode, this could also be used (used by many fields), in this case three useful products are also possible.
The “Smart network”: in this case there is no, or generally there isn’t, physical facility installed beside that would produce hydrogen by water dissolution beside the wind power plant, but the wind power plant produces energy straight to the network and maybe geographically further, on the spot of consumption the water dissolution capacity is placed (e.g.: hydrogen supply recharging wells), and through EPS-able intelligent solutions, maintaining the current stable status, controlled remotely produces hydrogen uses electricity, even at several different points of the country, in a decentralized way. The intelligent electric network is called “smart grid”, and it is not only suitable for the transmission and distribution of the electricity generated by wind power plants, but any other sustainable energy source (or not sustainable).
In reality, some wind hydrogen systems exist, for example the system located on the Norwegian Utsira island. The rapidly growing sustainable energy sources, EPS regulations and the question of energy storage are also raising important questions, and in the future hydrogen could also play a crucial role. It is especially true for Germany, where the installed wind power plant capacity is at around 20000 MW (note: this data was 55GW in 2020, see: https://www.cleanenergywire.org/factsheets/german-onshore-wind-power-output-business-and-perspectives), which will continue growing in the next couple of years, and the pumped storage plants or other solutions (e.g.. compressed air storage, CAES) will not be enough to solve the regulatory tasks.
The production of hydrogen using wind power or other renewable energy source is not an isolated task, where the only aim is to produce hydrogen, but it is a complex topic that fits into the currently existing energy infrastructure and cannot be treated with such important factors as intelligent networks.
Solar energy
Energy coming from the Sun – compared to human measures – is eternal and limitless: solar energy arriving to Earth is ~3.5*1024 J/year, which is about 17,000 times of the total annual energy needs of humanity. The problem coming from using solar energy is partly due to the seasons and time of days and the variants that come with it, which is made worse by the current weather conditions (e.g.: clouds). Nevertheless, it has a lot of advantages: it does not run out in the foreseeable future, it’s not polluting, does not need to be produced and transported, it does not get more expensive. Hydrogen production using solar energy is theoretically possible several ways:
- On of the most technologically mature methods is the production of electricity using photovoltaic (PV) method from solar energy, and eventually water decomposition. However, from an economic point of view, energy produced by photovoltaic systems is still very expensive, because of this the price of hydrogen produced using solar energy is also the highest.
- Another opportunity for the production of hydrogen using solar energy is the use of solar power plants. These focus direct solar radiation to a specific point using optical collector system, and thus produce a very high temperature. This temperature may reach above > 1500-2000°C, where the thermochemical decomposition of water (steam) occurs, namely to elements, hydrogen, and oxygen. The performance of the test solar power plant is 5kW- 80MW, but they can only operate 4-10 hours a day, they are quite expensive and involve risks.
- Another option for hydrogen production using solar power is photocatalysis, which is not yet widely known, but it’s an emerging method nowadays. How this method works is that certain catalysts are able to decomposition water using light, and therefore produce hydrogen. This fact has been known for decades that for example titanium dioxide is able to do that, but earlier photolysis was only carried out due to UV light. Nowadays, there are some promising research results about the development of multicomponent catalysts that are able to decomposition water economically, in the visible light range and a near room temperature state. Research aiming at photocatalyst water decomposition is carried out by the MTA Chemical Research Center. Please note that hydrogen can not only be retrieved from water using photocatalytic method, but from methanol as well.
Biomass
Biomass (that can include a lot of materials from agricultural waste, byproducts, products of energy plantations, to the sea algae population) could be a great hydrogen source in theory. Much like the fossil based hydrocarbons, biomass can be turned to hydrogen similarly by gasification or pyrolysis, which is followed by steam reforming. The advantage of this method could be that we possess extensive experience in the conversion and refining of fossil fuels.
Pyrolysis and gasification belong to the thermal procedures, but there are a few important differences, and none of them can be regarded similar to the direct burning of the fuel.
Traditional burning process appears in three things:
- flammable material (in this case: biomass),
- oxidizing material (in this case: the oxygen or air) and
The major difference is that in case of pyrolysis, oxygen is not present in the reaction (which happens between about 300-800°C), during gasification it is present, but its amount is not sufficient enough for full oxidization in the reaction (which occurs somewhere between 750-1600°C). Pyrolysis differs from burning by the latter being an exotherm process, meaning generating heat, the pyrolysis is endotherm, which means it needs heat intake for maintaining the process. The process of pyrolysis and gasification from organic materials (it cannot only be biomass, but organic waste as well or some other type of carbon hydrogen) in the first step has a high content of carbon monoxide (CO) and hydrogen (H2), results in a so called synthesis gas, which we usually reach using water steam to be able to produce more hydrogen from the process (this latter one means the above mentioned steam formation). During the process of pyrolysis, the oxygen present in biomass results in CO, while in other oxidation processes the oxygen intake is the main CO source. Besides hydrogen and CO, mostly carbon dioxide is produced in this process.
By burning biomass or pyrolysis theoretically only emits as much CO2 as the plant has maintained during its life. We also have to take into consideration that we need other input for production, for example mostly necessary fertilizers (which also requires a lot of hydrogen in form of ammonia), water and energy for production, harvesting and transportation, as well as other environmental aspects like the negative effects it had on the ground and biodiversity. Not mentioning the valuable agricultural lands’ reservation, if mainly – for energy purposes – as product biomass is produced, which might push out food value products. Due to all this we have to be careful about how much hydrogen we can get out of biomass (more specifically the intense energy focused production based biomass), or any other organic energy source (e.g.: biodiesel, bioethanol) production method, because it may easily cause environmental – or other societal or economic damage bigger than profit.
Waste
Municipal (or other organic) waste can also be used as a source for hydrogen, mainly using those methods, that we discussed above in the topic of biomass. Another possible solution is the anaerobic fermentation of waste, during which with the help of microorganisms produce biogas– in isolated circumstances from oxygen -, so highly methane containing gas (CH4). This methane is then – described at the traditional production methods using stream formation (SMR) – converted to hydrogen; the remaining biomass can be used as compost.
Biotechnological methods (Biohydrogen)
Certain unicellular organisms, e.g.: green algae or bacteria, can also deliver the process of the decomposition of water using the energy of the sun and produce hydrogen. For a while, Chlamydomonas Reinhardt has been known that the minerals produced during photosynthesis produce hydrogen in unfavorable conditions during a process called bio photolysis. There is only basic research on this field, but the aim is to build a facility that contains microorganisms, provide their reproduction, continuance, metabolism and produce hydrogen gas from these process as an end product. This photo bioreactor can produce cheap hydrogen in the future using the power of the sun. Research like this is also being carried out by the University of Szeged in Hungary.
Nuclear energy
As an alternative production method, hydrogen can be produced using nuclear energy. Using nuclear energy, high amounts of hydrogen can be produced in a centralized way. The following options are possible when it comes to hydrogen production using nuclear energy:
- thermochemical methods,
- high temperature electrolysis,
- hybrid processes.
(We evidently do not separately mention the “basic version”, the production using electricity provided by a nuclear power plant and forwarded in the network, through water decomposition hydrogen is produced.)
Storage, transportation, and application of Hydrogen
Storage of hydrogen in compressed gas and liquid state
Transportation of hydrogen in gas state is manageable in pipes: for example, there is a 25 bar pressured, 210 km long pipe network in the area of Ruhr for over 60 years. In comparison to natural gas pipes and racks, we have to take into consideration that hydrogen make steel brittle, due to its small size it diffuses easily. Storage of hydrogen can be managed in gas or liquid state. In reality storing hydrogen in gas state happens in high pressure barrels (4. image), but we have to pay special attention to avoid explosion danger.
Considerably high amount of hydrogen can be stored in cavities underground, in cultivated natural gas fields or in aquifers. Storage in liquid state makes a considerable amount of hydrogen storable but maintaining a temperature below -253°C is very costly, so it is only used in space technology. In the liquid storage hydrogen is always boiling as it arrives at the double walled barrels, so to avoid high pressure the extra pressure is always released. This results in the evaporation of liquid hydrogen in the long run, so it is only worth using this method in case high amounts of hydrogen is used. (Note: Mercedes, despite its difficulties has always been committed to storing hydrogen in liquid state in case fuel cell powered vehicle prototypes. In this topic Daimler Truck AG announced in December 2020 that cooperating with Linde they are developing a refill station suitable for liquid hydrogen pumping, which is needed for serving its planned manufacturing of GenH2 Trucks in 2023.)
High pressure storage usually occurs in two stages. First, hydrogen gas is pressured to a medium stage, or store it in liquid form, then after transfer compressors create the desired pressure. This is how it ends up in the trucks’ 350-700 bar pressure composite tanks.
At the topic of compressor, we have to mention that usual lubrication processes cannot be applied. (Note: Ionic compressors were developed and patented by Linde in 2006 which are pollution-free, low maintenance, highly effective compressors.)
Storage of hydrogen in bound state
Metal-hydride bottles absorb hydrogen in crystal structure, so they can store large amounts at low pressure. The most typically used materials are TiFex dust, which they fill into plain steel bottles. Nowadays, they are experimenting with nano pipes, which will hopefully be able to store 6 mass% hydrogen. In tradition high pressure bottles 2%, in the bottles presented in picture 6 3 mass% hydrogen can be stored.
Advantages of metal-hydride bottles:
- high energy density
- low pressure, so it can be refilled in current hydrogen infrastructure
- simple control
Disadvantages of metal-hydride bottles:
- can be filled slowly, from this aspect rather accumulator than a hydrogen storage
- hydrogen desorption speed is limited
- significant amount of heat is needed for desorption, and there is significant heat generated at refill
Metal-hydride bottles have to be continuously heated through hydrogen extraction, to be able to get all the amount of hydrogen. For this purpose, the heat generated by fuel cell is more than enough, but in case of cogeneration it may worsen the effectiveness of the system.
For years there have been researches about storing hydrogen in fullerene molecules. This carbon modification containing sixty atoms was discovered by Harry Kroto, Richard Smalleyand Robert Curl in 1985 in the University of Rice. With the discovery of the football sized molecule, the inventors won the chemical Nobel-prize in 1996. The “football” consists of 20 hexagons and 12 pentagons, this structure gives its stability. Its size doesn’t reach one nanometer, so the fullerene ball is one ten millionth of an actual football. It is only worth using it for storing hydrogen if it can observe at least 6 percent of its own mass. According to Boris Yakobson and his colleagues, hydrogen will remain in fullerene balls on room temperature in 8 percent density. ‘According to our calculations, certain fullerene balls can store hydrogen in such doses that it is similar to the density of metals” said Yakobson in an announcement posed by the University of Rice.
The bond among carbon atoms is one of the strongest chemical bonds. Yakobson’s team illustrated these bonds in a computer model, just like what happens to them if they include hydrogen atoms into the ball. Research claim that they can calculate the burst point of the fullerene ball with their model. This is important because that is the point when the nano bottle releases its content.
Transportation of hydrogen
One of the rudimentary questions of today is whether the distribution of hydrogen should be localized or centralized. It mostly depends on the production attributes of hydrogen (so we produce it in bigger factories or decentralized, several smaller locations), but similar to other fields of energy some sort of healthy balance of these can be realistic in the future. Depending on its geographical location local production and distribution is more suitable for certain applications. These could be for example refill stations far from the industrial hydrogen acquisition or hydrogen power supply for uninterrupted power supplies (UPS).
Nowadays, hydrogen is transported in compromised gas or liquid state, using several different ways of transportation. For the transportation of bigger amounts pipes are most suitable – based on the analogy of liquid methane – transportation on water, in case of smaller amounts traffic and rail transport seem to be the most suitable solutions.
Nowadays, the most frequently used method for transportation of hydrogen is traffic, since most consumers need considerably low amount, and in these cases production on location would be too costly. There are two ways to transport hydrogen in traffic: compromised in bigger bottles or in liquid state in barrel cars. As we stated earlier, production and storage of compromised hydrogen gas is easier and more efficient than liquid. But during transportation small energy density has its downsides: one truck can carry around 500 kg hydrogen in 200 bar bottles. Compared to the 40 tons of the trucks this usage seems fairly low.
For the transportation of hydrogen in liquid form, double-walled, vacuumed barrel cars are used, which are transported to the consumer by trucks or trains. With these, 4000 kg hydrogen can be transported – which is the equivalent of 13,000 liter of petrol. Liquid state can be transported on water in theory, if the hydrogen economy reaches a level that the production of consumption of hydrogen will be divided by great distances. In this area experience gathered through the transportation of LNG (fluid natural gas) can be of great help.
Consumers in need of high amount of hydrogen (e.g.: chemical or petrochemical factories) can be supplied optimally – and in case the circumstances are given – through pipe systems between pressure of 10-70 bar. Due to the growing need of industrial consumers in the last years, hydrogen pipelines have been dynamically extended – mostly directly between the producers and the great consumers – since this is the most economic form of transportation.
Due to the previously mentioned attributes of hydrogen (fugacity, material weakening, material exhaustion) for the transportation of hydrogen special steel compounds or other composite materials are needed, which is also true for extensions, seals, and aiding tools as well for example compressors. This evidently affects investment budget, so building a hydrogen pipe system is more expensive than of for example natural gas.
Several hydrogen pipes are in operation even today. In Germany, for example a 240 kilometer long, 30 bar pressure network supplies its consumers in the Ruhr region since the 1930s. In the EU, the summary of hydrogen pipes is 1600 km (half of this is located in Belgium and the Netherlands). In Kazincbarcika, on the premises of BorsodChem some kilometer long hydrogen pipe system delivers the gas from its production point (HyCo factories) to its consumption location. In the USA and Canada all together there is about 1800 km already existing, industrial hydrogen pipe system in operation safely for decades.
According to some opinions the most modern natural gas pipelines will be suitable for hydrogen transportation as well, but this for now remains debatable. It is still being argued and researched whether it is possible to introduce small percentage of hydrogen into the natural gas network, would it cause any damage to the transportation system or in the devices of the end consumer.
Future energetic application of hydrogen
We have arrived at the edge of hydrogen being used for energetic purposes, and there is a great chance that hydrogen and hydrogen energetic technologies will gain significant share in the upcoming decades. Energetic purpose consumption, including transportation, appeared in 2000s, since projects for fuel cell busses or passenger cars were successfully carried out in many countries, several hydrogen refill stations build, and early demonstrational projects in the field of energy production were in place. Nonetheless, they served the purpose of technological validation and testing; the economic aspects or commercialization (mass production for the market) were not yet given, except for special applications.
Source: Mayer Zoltán és Kriston Ákos – Hidrogén és metanol gazdaság (https://regi.tankonyvtar.hu/hu/tartalom/tamop412A/2010-0017_31_hidrogen_es_metanol/ch01s04.html)
Fuel cells
At a Hannover Messe in 2003, a completely ecofriendly energy producing equipment caught our attention presented in the new energetics solutions area of the exhibition. The item in question is a fuel cell, a electrochemical converter, which produces electricity highly effectively with the catalytic reaction of hydrogen and oxygen. The produced byproduct is water.
It had been an old and a continuous aim of our company to present more and more of its own products among its activities. After our visit to the Hannover fair, we decided this new technology was worth learning more about. Even though the history of fuel cells goes back to 1839 to the discovery made by William Grove, it was only in the last two decades that their mass application has come to spotlight.
Get in KONTAKT
We are glad to support you with our services in the areas of electrical manufacturing, design, consulting, development, robotics, or sensor technology.